
BENZA BLOCK S PREMIUM DX
ベンザブロックSプレミアムDX
CHARACTERISTICS
- d-Chlorpheniramine maleate, an antihistamine, relieves runny nose, stuffy nose, and sneezing.
- Pseudoephedrine hydrochloride reduces stuffy nose by relieving swelling and hyperemia of the nasal mucosa.
- Belladonna total alkaloids suppress runny nose by blocking the transmission of stimuli to the nasal glands.
- Contains 9 ingredients to relieve various cold symptoms.
- Easy-to-take, small, light-yellow caplet.
USES
Relief of various symptoms of a cold: runny nose, stuffy nose, sore throat, sneezing, cough, phlegm, feeling cold (chills due to fever), fever, headache, joint pain, and muscle pain.
DOSAGE AND DIRECTIONS
| Age | One dose | Daily dose |
| 15 years or over | 2 tablets | 3 times |
| Under 15 years | Do not take | |
Precautions regarding dosage and directions
- Please follow the recommended dosage and directions.
- How to take out a caplet
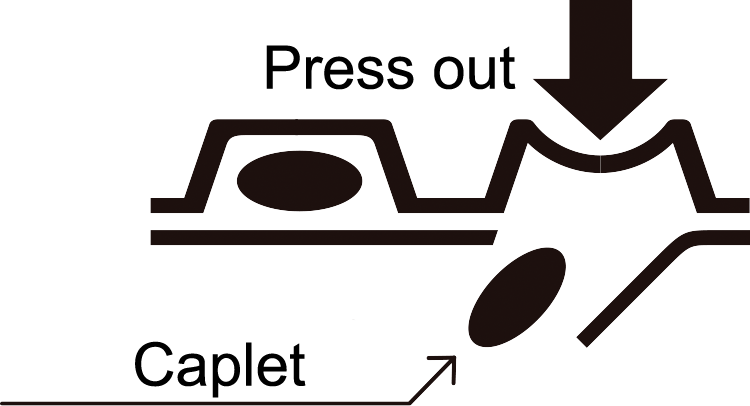
As shown in the figure, press the protruded portion of the PTP sheet with your finger to tear the aluminum foil and take out a caplet. (If you swallow the PTP sheet by mistake, its sharp edges may perforate your esophagus, etc., resulting in serious complications.)
INGREDIENTS
In 6 tablets (daily dose)
| ACTIONS | INGREDIENTS | CONTENTS |
|---|---|---|
| Reduces fever and relieves pain | Acetaminophen | 900mg |
| Relieves runny nose and sneezing | d-Chlorpheniramine maleate | 3.5mg |
| Relieves stuffy nose | Pseudoephedrine hydrochloride | 135mg |
| Relieves runny nose | Belladonna total alkaloids | 0.3mg |
| Relieves cough | Dextromethorphan hydrobromide hydrate | 48mg |
| Promotes sputum expectoration | Bromhexine hydrochloride | 12mg |
| Relieves sore throat | Tranexamic acid | 420mg |
| Relieves headache | Anhydrous caffeine | 75mg |
| Vitamin | Riboflavin (vitamin B2) | 12mg |
Inactive ingredients:
Cellulose, corn starch, croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, partially hydrolyzed polyvinyl alcohol, hypromellose, talc, titanium oxide, red ferric oxide
Precautions regarding ingredients:
Although urine may turn yellow after taking this medicine, do not worry because this is caused by riboflavin in this medicine.
PRECAUTIONS FOR USE
What you should NOT do
(If you do not follow the precautions, it may worsen your symptoms, or you may be at an increased risk of side effects and accidents.)
- The following persons should not take this medicine:
- Persons who have experienced allergic reactions to this medicine or any of its ingredients.
- Persons who have experienced asthma after taking this medicine or other cold medications or antipyretic analgesics.
- Persons who have suffered from the following symptoms: Dysuria due to prostatic hypertrophy
- Persons diagnosed with the following conditions: Hypertension, heart disease, thyroid disorders, diabetes
- Do not use any of the following medications while you take this medicine:
Other cold medicines, antipyretic analgesics, sedatives, cough and expectorant medicines, and oral medicines containing antihistamines (including oral medicines for rhinitis, motion sickness medicines, allergy medicines, and sedative‑hypnotics), gastrointestinal analgesics and antispasmodics, and oral medicines containing tranexamic acid - Do not operate vehicles or machines after taking this medicine. (Symptoms such as sleepiness, blurred vision, and abnormal glare may appear.)
- Do not drink alcohol before or after taking this medicine.
- Do not use this medicine continuously for a long period.
Who should seek consultation
- The following persons should consult a physician, pharmacist, or registered salesperson before taking this medicine:
- Persons currently receiving treatment from a physician or dentist.
- Women who are or may be pregnant.
- Breastfeeding women.
- Elderly persons.
- Persons who have experienced allergic reactions to medicines or other substances.
- Persons with the following symptoms: high fever, difficulty in urination.
- Persons diagnosed with the following conditions: Liver diseases, kidney diseases, gastroduodenal ulcer, glaucoma, persons with blood clots (cerebral thrombosis, myocardial infarction, thrombophlebitis), persons at risk of developing thrombosis.
- Persons on treatment with monoamine oxidase inhibitors (such as selegiline hydrochloride).
- Persons who have experienced insomnia, dizziness, feeling of weakness, shaking, or palpitations due to cold medicines, cough and expectorant medicines, oral medicines for rhinitis, etc.
- Persons taking oral remedies containing parasympatholytic agents. (belladonna total alkaloids, isopropamide iodide, scopolia extract, etc.)
- Stop taking this medicine immediately and consult a physician, pharmacist, or registered salesperson if the following symptoms appear after taking this medicine because they might be side effects of the medicine. Take this leaflet with you.
Affected area Symptoms Skin Rashes, redness, itching Digestive organs Nausea, vomiting, loss of appetite Neuropsychiatric system Dizziness, convulsions, headache, insomnia, nervousness Respiratory system Shortness of breath, breathing difficulties Urinary organs Difficulty in urination Others Excessive decrease in body temperature, facial flushing, abnormal glare In rare cases, serious symptoms might occur. In such a case, immediately consult a physician.
Name of symptoms Symptoms Shock
(Anaphylaxis)Symptoms such as itching of the skin, urticaria, hoarseness, sneezing, itching of the throat, breathing difficulties, palpitations, and clouded consciousness will appear immediately after taking the medicine. Mucocutaneous ocular syndrome (Stevens-Johnson syndrome), toxic epidermal necrolysis, acute generalized exanthematous pustulosis Symptoms such as high fever, hyperemia of the eyes, eye discharge, ulceration of the lips, sore throat, rashes and redness of a large area of the skin, small rashes (small pustules) on reddened skin, heaviness of the whole body, and no appetite persist or suddenly worsen. Drug-induced hypersensitivity syndrome Symptoms such as widespread skin redness, generalized rash, fever, general malaise, and swelling of lymph nodes (in the neck, armpits, groin, etc.) Hepatic function disorder Symptoms such as fever, itching, rashes, jaundice (yellowing of the skin and white part of the eye), brown urine, heaviness of the whole body, and loss of appetite will appear. Renal disorder Symptoms such as fever, rashes, reduced urine output, swelling of the whole body, heaviness of the whole body, joint pain (feeling pain in the joints), and diarrhea will appear. Interstitial pneumonia When you go upstairs or exert yourself too much, symptoms such as shortness of breath, breathing difficulties, dry cough, and fever will appear. They will appear suddenly and persist. Asthma Symptoms such as wheezing during breathing and breathing difficulties will appear. Hypoplastic anemia Symptoms such as bruising, nose bleeding, bleeding gums, fever, pale skin or membranes, feeling of fatigue, palpitations, shortness of breath, feeling ill, dizziness, and blood in urine will appear. Agranulocytosis Symptoms such as sudden high fever, chills, and sore throat will appear. - Stop taking this medicine and consult a physician, pharmacist, or registered salesperson if the following symptoms appear after taking this medicine and continue or worsen. Take this leaflet with you.
Constipation, dry mouth, sleepiness, blurred vision, diarrhea. - Stop taking this medicine and consult a physician, pharmacist, or registered salesperson if symptoms do not improve after taking this medicine five to six times. Take this leaflet with you.
PRECAUTIONS FOR STORAGE AND HANDLING
- Store the medicine in its box in a dry, cool place away from direct sunlight.
- Keep out of the reach of children.
- To prevent improper use or loss of quality, do not transfer the medicine to another container.
- Do not take this medicine after the expiration date.
- Fill in the date when the inner pouch (aluminum pouch) was opened in the column provided on the box.
- Once the inner pouch (aluminum pouch) is opened, take this medicine within about 6 months to maintain quality.
